Sea surface temperature at record high
The image below, created with Climate Reanalyzer screenshots, shows that the sea surface temperature (60°S - 60°N mean) was 21.2°C on April 20, 2024, a record high, and slightly higher than the record high temperatures reached earlier this year.The image below, adapted from NOAA, shows global ocean temperature anomalies from 1901-2000. The green line (LOcally Estimated Scatterplot Smoothing) could be a warning of higher temperature anomalies to come.
The image below, adapted from Copernicus, shows March 2024 sea surface temperature anomalies from 1991-2020. High anomalies show up, especially around the Equator which can be expected at this time of year.
North Atlantic heating up
The North Atlantic Ocean is heating up, as more sunlight starts reaching the Northern Hemisphere. The image below, adapted from Climate Reanalyzer, shows sea surface temperatures up to April 11, 2024. The image shows that 2024 temperatures have been significantly higher than 2023 temperatures for the same date. The annual maximum temperature in 2023 was reached on August 31. Temperatures can be expected to rise dramatically over the next few months, in line with the change in seasons.
Much will depend on the strength of the current El Niño over the next few months. The image below, adapted from NOAA, shows monthly temperature anomalies versus 1901-2000 through March 2024, colored by El Niño/La Niña conditions.
NOAA warns that there is a bit of a delay in the effects of any given ENSO phase. So, the first part of this year will still be influenced by El Niño, which is in part why NOAA predicts a 55% chance that 2024 will be hotter than 2023.
Further factors (other than El Niño) may continue to accelerate the temperature rise, as discussed in earlier posts such as this one. One danger is that, due to strong wind along the path of the Gulf Stream, huge amounts of ocean heat will abruptly get pushed into the Arctic Ocean, with the influx of ocean heat causing destabilization of hydrates contained in sediments at the seafloor of the Arctic Ocean, resulting in eruptions of huge amounts of methane, as discussed in earlier posts such as this one.
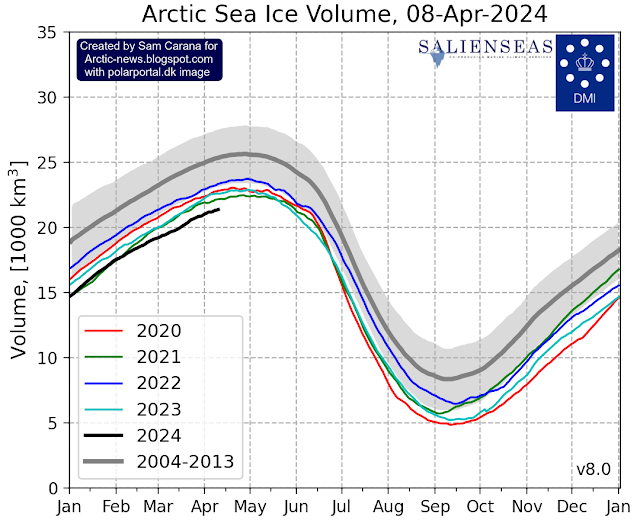
Arctic sea ice getting very thin
Given that Arctic sea ice currently is still relatively extensive, this low volume indicates that sea ice is indeed very thin, which must be caused by ocean heat melting sea ice from below, since little or no sunshine is yet reaching the Arctic at the moment and air temperatures are still far below freezing point, so where ocean heat may be melting sea ice away from below, a thin layer of ice will quickly be reestablished at the surface.
This situation looks set to dramatically change over the next few months, as air temperatures will rise and as more ocean heat will reach the Arctic Ocean. Moreover, as illustrated by the map below, much of the thicker sea ice is located off the east coast of Greenland. This sea ice and the purple-colored sea ice can be expected to melt away quickly with the upcoming rise in temperatures over the next few months.
Climate Emergency Declaration
The situation is dire and the precautionary principle calls for rapid, comprehensive and effective action to reduce the damage and to improve the situation, as described in this 2022 post, where needed in combination with a Climate Emergency Declaration, as discussed at this group.
• Climate Reanalyzer
https://climatereanalyzer.org
• NOAA - Ocean temperature anomalies
https://www.ncei.noaa.gov/access/monitoring/climate-at-a-glance/global/time-series/globe/ocean/1/0/2015-2024?filter=true&filterType=loess
• Copernicus sea surface temperature anomalies
https://pulse.climate.copernicus.eu
• NOAA - Monthly Temperature Anomalies Versus El Niño
https://www.ncei.noaa.gov/access/monitoring/monthly-report/global/202403/supplemental/page-4
• NOAA - ENSO update
https://www.facebook.com/NOAAClimateGov/posts/821505663344434
also discussed at facebook at: https://www.facebook.com/groups/arcticnews/posts/10161353804294679
https://arctic-news.blogspot.com/2024/04/did-the-climate-experience-a-regime-change-in-2023.html
• Atlantic ocean heat threatens to unleash methane eruptions
https://arctic-news.blogspot.com/2024/03/atlantic-ocean-heat-threatens-to-unleash-methane-eruptions.html
https://seaice.uni-bremen.de/start
• Danish Meteorological Institute - Arctic sea ice thickness and volume
https://ocean.dmi.dk/arctic/icethickness/thk.uk.php
• Transforming Society
https://arctic-news.blogspot.com/2022/10/transforming-society.html
• Climate Plan
https://arctic-news.blogspot.com/p/climateplan.html
• Climate Emergency Declaration
https://arctic-news.blogspot.com/p/climate-emergency-declaration.html